Effects of Penicillin
What Penicillin does- Penicillin is bactericidal [5]. It works by inhibiting bacterial cell wall synthesis, resulting in eventual lysis of the cell.
Types of Penicillin- There are over 50 types of antibiotics classified as penicillin [5]. Of all the different types of penicillin, there are two types that are more commonly used than the others: penicillin G and penicillin V.
This section will cover what each of the types can treat, chemical differences, regular dosage, and side effects for each.
Types of Penicillin- There are over 50 types of antibiotics classified as penicillin [5]. Of all the different types of penicillin, there are two types that are more commonly used than the others: penicillin G and penicillin V.
This section will cover what each of the types can treat, chemical differences, regular dosage, and side effects for each.
Penicillin G

Penicillin G, also known as benzylpenicillin (IUPAC name: (2S,5R,6R)-3,3-dimethyl-7-oxo-6-(2-phenylacetamido)-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid [3]), is typically given prenatally because it is very active and is not stable in the hydrochloric acid of the stomach. Structurally, the R group is taken by a benzyl group.
Penicillin G is typically prescribed as a potassium salt, where the hydrogen of the carboxylic acid is replaced by a potassium group. Penicillin G is prescribed as an injection and is used for serious infections, as compared to Penicillin V. [4]
Penicillin G is typically prescribed as a potassium salt, where the hydrogen of the carboxylic acid is replaced by a potassium group. Penicillin G is prescribed as an injection and is used for serious infections, as compared to Penicillin V. [4]
|
Used to Treat-
Streptococcus pyogenes: Pneumonia, empyema, pericarditis, endocarditis, meningitis - Bacillus anthracis: Anthrax - Actinomyces israelii: Actinomyocosis - Clostridium: Botulism - Corynebacterium diphtheria: Diphtheria - Erysipelothrix rhysiopathiae: Erysipelothrix endocarditis - Fusobacterium and spirochetes: Fusospirochetosis - Listeria monocytogenes: Meningitis and endocarditis - Neisseria gonorrhoeae: Disseminated gonococcal infections - Treponema pallidum: Syphilis (12-24 million units a day) - Neisseria meningitidis: Meningococcal meningitis |
Dosages
12 to 24 million units per day divided between every 4-6 hours 5 to 24 million units a day divided between every 4-6 hours Minimun of 8 million units/day divided in doses every 6 hrs 20 million units a day 2-3 million units/day for 10-12 days 12-20 million units a day for 4-6 weeks 5-10 million units a day 15-20 million units a day for 2-4 weeks 10 million units a day 12-24 million units a day 24 million units/day as 2 million units per 2 hours |
Side Effects:
- Upon initial start of penicillin G injections, a Jarisch-Herxheimer reaction may occur systemically that causes fever, chills, headache, myalgias, tachycardia, hyperventilation, and vasodilation with hypotension. These side effects can disappear within 12-24 hours.
- Hypersensitivity
- GI Effects: Nausea, vomiting, black or hairy tongue, stomatitis
- Hematological: Neutropenia, Hemolytic anemia
- Metabolic: Hyperkalemia
- Nervous system: Hyperreflexia, twitches, seizures, and coma.
- Urogenital: Interstitial nephritis, hematuria.
- Upon initial start of penicillin G injections, a Jarisch-Herxheimer reaction may occur systemically that causes fever, chills, headache, myalgias, tachycardia, hyperventilation, and vasodilation with hypotension. These side effects can disappear within 12-24 hours.
- Hypersensitivity
- GI Effects: Nausea, vomiting, black or hairy tongue, stomatitis
- Hematological: Neutropenia, Hemolytic anemia
- Metabolic: Hyperkalemia
- Nervous system: Hyperreflexia, twitches, seizures, and coma.
- Urogenital: Interstitial nephritis, hematuria.
Penicillin V
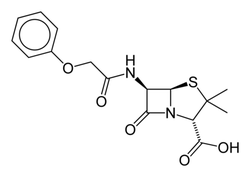
Penicillin V, also known as phenoxymethylpenicillin (IUPAC name: 3,3-dimethyl-7-oxo-6-(2-phenoxyacetamido)-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid)[1], is a commonly used administered variation of penicillin. Structurally, the main difference in penicillin V is that it contains (for its R group) a methyl group, attached to an oxygen followed by a phenyl group. [2]
Penicillin V is often prescribed in a form of penicillin called "Penicillin VK," where the potassium group replaces the proton on the carboxylic acid. Penicillin V is a less active, and a less potent version of penicillin G. Because of these properties, Penicillin V may be administered orally, but can treat a smaller number of bacteria, as well as less potent bacteria in general.
Penicillin V is often prescribed in a form of penicillin called "Penicillin VK," where the potassium group replaces the proton on the carboxylic acid. Penicillin V is a less active, and a less potent version of penicillin G. Because of these properties, Penicillin V may be administered orally, but can treat a smaller number of bacteria, as well as less potent bacteria in general.
|
Used to Treat:
- Staphylococcal infections - Pneumococcal infections that are mild to severe located in the upper respiratory tract - Streptococcal infections: infections in the respiratory tract, scarlet fever, or mild erysipelas - Fusospirochetosis (Gingivitis) |
Dosages:
250-500mg every 6 to 8 hours 250-500mg every 6 to 8 hours 125-250mg every 6 to 8 hours for 10 days 250-500mg every 6 to 8 hours |
[1] N.p. Web. 14 Apr 2013. <http://www.drugbank.ca/drugs/DB00417>.
[2] RxList, N.p. Web. 14 Apr 2013. <http://www.rxlist.com/penicillin-vk-drug.htm>.
[3] N.p. Web. 14 Apr 2013. <http://www.drugbank.ca/drugs/DB01053>.
[4] "Penicillin G (Pfizerpen)." Medify. N.p., n.d. Web. 14 Apr. 2013."Penicillin G Potassium (Penicillin G Potassium) Drug Information: Description, User Reviews, Drug Side Effects, Interactions - Prescribing
[5] Donald J. Tipper and Jack L. Strominger. "Mechanism of Action of Penicillins: A Proposal Based on their Structural Similarity to Acyl-D-alanyl-D-alanine." Proceedings of the National Academy of
Sciences of the United States of America. 1965. Pages 1133-1141. Information at RxList."RxList.
[6] Streble, Susan. "The Evolution of Resistance to Penicillin." Allegheny College: Webpub. 2001.
Image source 1: http://en.wikipedia.org/wiki/File:Penicillin-G.svg
Image source 2: http://en.wikipedia.org/wiki/File:Penicillin-V-2D-skeletal.png
Image source 3: http://en.wikipedia.org/wiki/File:Procaine.svg
Image 4 source: http://dermatlas.med.jhmi.edu/data/images/black_hairy_tongue_1_040620.jpg
[2] RxList, N.p. Web. 14 Apr 2013. <http://www.rxlist.com/penicillin-vk-drug.htm>.
[3] N.p. Web. 14 Apr 2013. <http://www.drugbank.ca/drugs/DB01053>.
[4] "Penicillin G (Pfizerpen)." Medify. N.p., n.d. Web. 14 Apr. 2013."Penicillin G Potassium (Penicillin G Potassium) Drug Information: Description, User Reviews, Drug Side Effects, Interactions - Prescribing
[5] Donald J. Tipper and Jack L. Strominger. "Mechanism of Action of Penicillins: A Proposal Based on their Structural Similarity to Acyl-D-alanyl-D-alanine." Proceedings of the National Academy of
Sciences of the United States of America. 1965. Pages 1133-1141. Information at RxList."RxList.
[6] Streble, Susan. "The Evolution of Resistance to Penicillin." Allegheny College: Webpub. 2001.
Image source 1: http://en.wikipedia.org/wiki/File:Penicillin-G.svg
Image source 2: http://en.wikipedia.org/wiki/File:Penicillin-V-2D-skeletal.png
Image source 3: http://en.wikipedia.org/wiki/File:Procaine.svg
Image 4 source: http://dermatlas.med.jhmi.edu/data/images/black_hairy_tongue_1_040620.jpg

